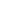
The Paul glaucoma implant primarily comprises a plate and a tube, with an adhesive layer situated between the tube's exterior and the ridge groove's interior. Its plate boasts a surface area of 342.1mm². Strategically placed window holes within the plate enable fiber insertion and secure fixation to the eye. The tube effectively channels aqueous humor from the anterior chamber to the eye's exterior, while the adhesive securely bonds the tube within the plate's ridge groove.
**Working Principle:** The Paul glaucoma implant functions by maintaining the patency of the surgical drainage opening, preventing healing and closure to enhance glaucoma surgery outcomes. Most implants incorporate a fluid-conducting tube; the remaining solid structure facilitates aqueous outflow along the implant surface. Regardless of design, all implants share a common objective: reducing intraocular pressure (IOP) by augmenting aqueous humor drainage.
Glaucoma implants trace their evolution to the Molteno implant, first trialed in 1969. Following the China-approved Ahmed glaucoma implant, numerous other designs have emerged.
Related Products
Department: Dermatology
Department: Dermatology
Department: Dermatology
Department: Dermatology
The Cochlear™ Nucleus® Profile Series C1632 Cochlear Implant
Related Packages
SPOT-MAS Package (Cancer Look Out)




Stem Cell Therapy for Arthritis




Fecal Microbiota Transplantation, FMT


Painless Gastrointestinal Endoscopy




Heart & Head & Neck Coronary CTA Package